Lay Summary
PIGO-CDG, also known as PIGO deficiency, is a rare inherited condition that affects several body systems. There have been 18 cases of PIGO-CDG reported to date in the medical literature. PIGO-CDG is classified as a disorder of GPI anchor biosynthesis. PIGO-CDG is caused when an individual has mutations in both copies of their PIGO gene which provides instructions for making a protein that participates in building GPI anchors. GPI anchors are molecules that “anchor” certain proteins to the cell surface. The PIGO protein is involved in one of the final steps of GPI anchor synthesis where it attaches a molecule called ethanolamine phosphate to the end of the forming GPI anchor. Mutations in the PIGO gene cause defects in GPI-anchored proteins which make them unstable or unable to attach to the surface. Symptoms of PIGO-CDG begin in infancy and the clinical presentation is primarily characterized by developmental delay, hypotonia, intellectual disability, seizures, abnormal facial features, cardiac abnormalities and unusually short length of fingertips. Stunted growth and gastrointestinal issues may also be present in some individuals with this disorder. Individuals diagnosed with PIGO-CDG have variable life expectancies, with prognoses depending on the severity of symptoms. PIGO-CDG is usually diagnosed through genetic testing, however, testing for the presence of GPI-anchored proteins on certain blood cells can also identify PIGO-CDG. Except for ketogenic diets which may reduce seizures, there are currently no approved treatments for PIGO-CDG. While gene replacement therapy is being explored, current treatment is focused on the management of specific symptoms and preventing complications.
Overview
Phosphatidylinositol glycan class O congenital disorder of glycosylation (PIGO-CDG) is a rare autosomal recessive genetic disorder. The first reported case of PIGO-CDG was in 2012, and 18 confirmed cases have been reported in the literature to date 1–8. PIGO-CDG is commonly referred to as PIGO deficiency or Mabry syndrome.
The PIGO gene encodes a subunit of an enzyme complex that is involved in glycosylphosphatidylinositol (GPI) anchor biosynthesis. The enzyme complex, comprised of PIGO and PIGF proteins, catalyzes the transfer of ethanolamine phosphate to the third mannose residue in the GPI core. GPI anchors are an important mechanism of attachment for proteins to the cell surface, and these proteins are known as GPI-anchored proteins. Mutations in PIGO result in defects in GPI-anchored proteins, which are critical to the development and function of many bodily systems, including the nervous system.
Symptoms of PIGO-CDG begin in infancy and the characteristic presentation includes developmental delay, seizures, facial abnormalities, unusually short fingertips, genital abnormalities, gastrointestinal problems, as well as psychomotor impairments 1–8. Although a definitive diagnosis can only be achieved through genetic sequencing, analyzing blood cells for an absence of GPI-anchored proteins can help diagnose disorders of GPI-anchor biosynthesis. No treatment is currently available for PIGO-CDG, however, gene therapy is a potential therapeutic strategy9.
Synonyms
- Hyperphosphatasia with mental retardation syndrome 2 (HPMRS2)
- Mabry Syndrome
- Phosphatidylinositol glycan class O congenital disorder of glycosylation
- GPI biosynthesis defect type 6 (GPIBD6)
- PIGO deficiency
Inheritance
PIGO-CDG is an autosomal recessive disorder, meaning an affected individual inherits one defective copy of the gene from each asymptomatic parent.
Gene Function
The PIGO gene encodes the catalytic subunit (PIG-O) of the GPI ethanolamine phosphate transferase III (GPI-EtNP transferase III) complex.
The GPI-EtNP III enzyme is located in the endoplasmic reticulum (ER) where it catalyzes the transfer of ethanolamine phosphate to the third mannose residue of the GPI anchor core which facilitates the attachment of the GPI-anchor to the C-terminus of proteins10.
GPI-Anchored Protein Biosynthesis
GPI-anchored protein biosynthesis is one of the major glycosylation pathways that attach glycans to lipid molecules within cells. Many proteins are attached to the cell surface by GPI anchors, which are referred to as GPI-anchored proteins.
The core structure of GPI consists of phosphatidylinositol (PI), glucosamine (GlcN), three mannose sugars (Man3), and phosphoethanolamine (EtN-P) connected to each other in that sequence (Figure 1). GPI-anchored proteins are attached to the GPI by forming a bond between the EtN-P group of the GPI core and the C-terminus of newly synthesized proteins in the ER lumen 10.
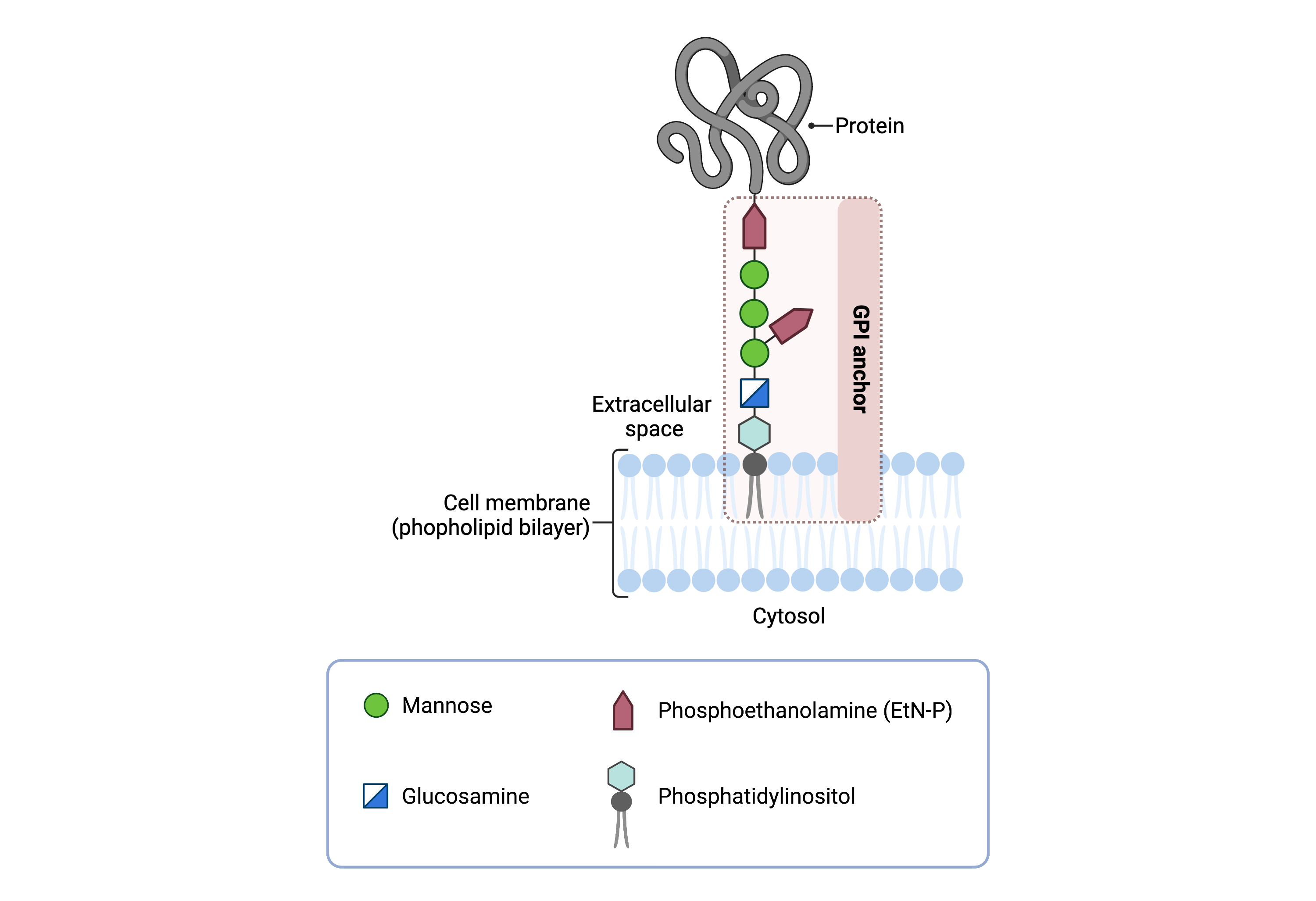
Figure 1. Overview of GPI-anchored protein structure.
The GPI anchor is added to proteins, generating GPI-anchored proteins. The GPI anchor section lodges into the membrane, attaching the protein to the membrane. The GPI anchor involves phosphatidylinositol, glucosamine, mannose and phosphoethanolamine.
The generation of GPI-anchored proteins is a multi-step process involving more than 30 enzymes and can be divided into the following steps (Figure 2):
- GPI anchor synthesis
- Protein attachment
- Lipid/glycan remodelling and protein transport
GPI anchor synthesis and protein attachment are largely carried out by a series of enzymes encoded by the PIG genes, while enzymes encoded by the PGAP genes facilitate remodelling of the GPI-anchored protein.
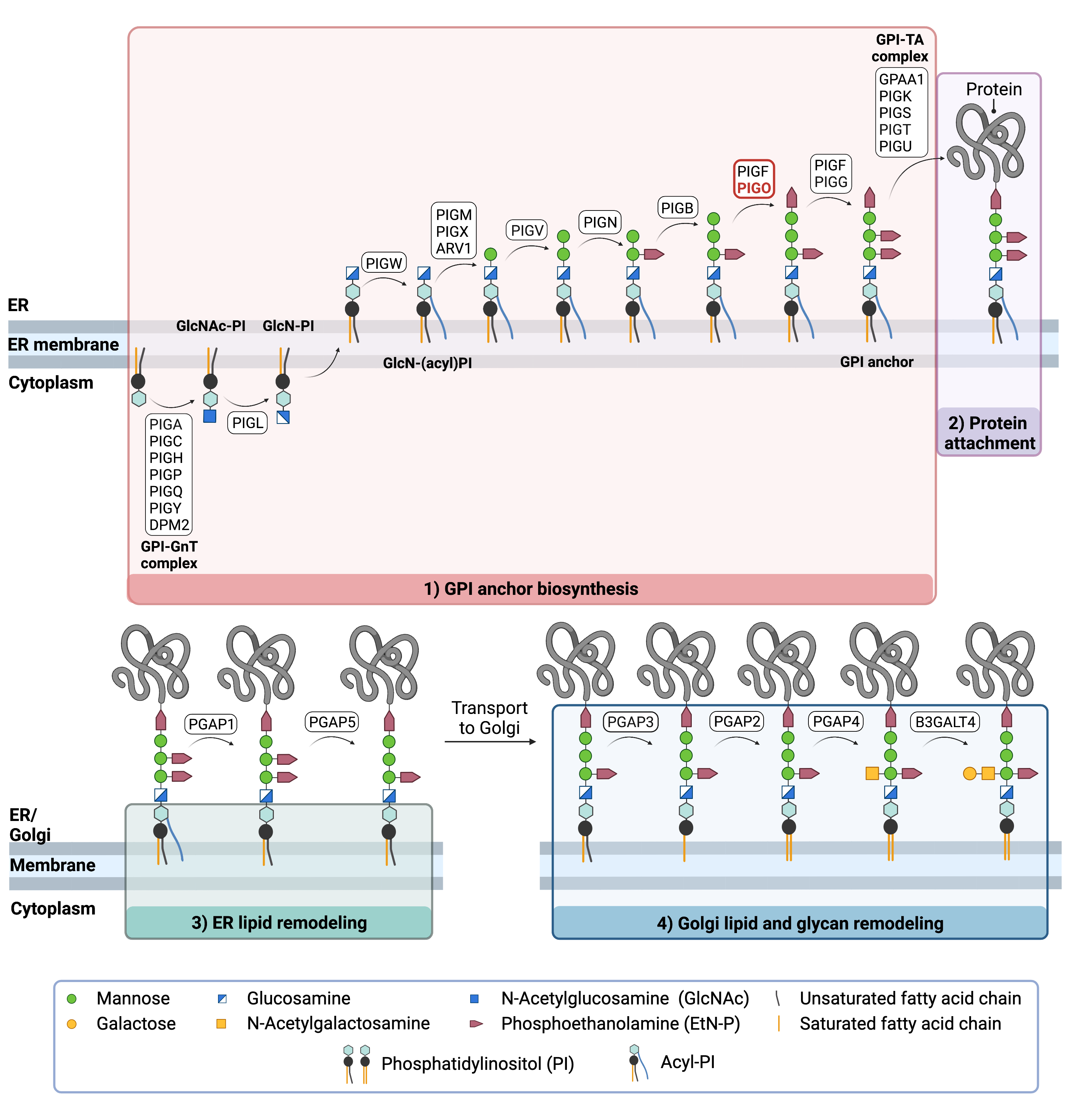
Figure 2. Overview of GPI-anchored protein biosynthesis and the role of PIGO.
GPI-anchored protein biosynthesis relies on a series of enzymatic reactions. First, the GPI anchor core is built on the ER membrane, where enzymes modify the lipid and glycan portions, generating PI-GlcN-Man3-EtN-P. The protein is then attached to the GPI anchor in the ER, before further modifications are made to the lipid and glycan portions in the ER and the Golgi. PIG-O is a subunit of the enzyme complex that catalyzes the transfer of phosphoethanolamine (EtN-P) to the third mannose of the GPI anchor core which facilitates the attachment of the GPI anchor to proteins.
GPI Anchor Synthesis
The first stage of GPI anchor biosynthesis involves the stepwise construction of the GPI anchor. The first step involves the transfer of the monosaccharide N-acetylglucosamine (GlcNAc) from nucleotide sugar UDP-GlcNAc to the lipid phosphatidylinositol (PI), generating GlcNAc-PI. The transfer of GlcNAc is catalyzed by GPI GlcNAc transferase (GPI-GnT), a 7-subunit protein complex 10–12:
GlcNAc-PI then has its acetyl (Ac) group removed from the GlcNAc sugar, generating glucosamine phosphatidyl inositol (GlcN-PI) which is transported into the luminal side of the ER19,21. Within the ER, a fatty acid chain is attached to GlcN-PI, generating GlcN-(acyl)PI. The final steps of the GPI anchor synthesis involve the sequential addition of 3-4 mannose sugars and three EtN-P groups by several PIG enzymes. Together PIG-O and PIG-F form the GPI-ethanolamine phosphate transferase III complex where PIG-O is the catalytic subunit 13. This enzyme complex catalyzes the attachment of the second ethanolamine phosphate (EtN-P) to the third mannose residue of the GPI core. After the third EtN-P group is attached, the completed GPI anchor core, PI-GlcN-Man3-EtN-P, is ready to be attached to a protein 10–12.
Protein Attachment
Once the GPI anchor has been synthesized, it is transferred en bloc to a protein with a C-terminal GPI attachment signal sequence by GPI transamidase (GPI-TA). The five-protein enzyme complex catalyzes the simultaneous cleavage of the signal sequence and attachment of the GPI anchor to the newly synthesized protein 10–12.
Lipid/Glycan Remodelling and Protein Transport
After the protein has been attached to the GPI anchor, both the glycan and lipid portion of the anchor undergo modifications (referred to as remodelling) in the ER and Golgi by post-GPI attachment to protein (PGAP) enzymes or the glycosyltransferase B3GALT4. The complete GPI-anchored protein is then transported to the plasma membrane where it associates with other GPI-anchored proteins in lipid rafts 14.
Disease Mechanism
Mutations in the PIGO gene results in impaired stability and function of the GPI-EtN-P transferase III enzyme, which is essential for enabling the attachment of GPI anchors to proteins. This results in the failure to complete the GPI biosynthesis pathway and an impaired ability of GPI anchors to bind proteins, leading to a reduction in functional GPI anchored proteins on the cell surface8. GPI-anchored proteins have several critical functions in the cell such as adhesion molecules, receptors, and enzymes in signal transduction pathways, and are also important in embryogenesis and neurogenesis5.
Mutations
The PIGO gene is located on chromosome 9 (9p13). To date, 13 variants in the PIGO gene have been reported, with patients typically presenting with nonsense/frameshift mutation in one allele, resulting in no PIGO functionality, and a missense mutation in the other allele, which may have residual PIGO activity 5–7. Therefore, the severity of the disease may depend on the level of residual activity left in the PIGO allele that has a missense mutation. The Arg119Trp mutation has been reported to have a more severe phenotype in PIGO-CDG patients7.
Signs and Symptoms
Clinical Presentation
Individuals with PIGO-CDG typically develop signs and symptoms during infancy. PIGO-CDG is primarily characterized by intellectual disability, hypotonia epilepsy, abnormal facial features and anorectal abnormalities. Common symptoms of PIGO-CDG include1–8 :
- Neurological– global developmental delay, intellectual disability, low muscle tone (hypotonia), and epileptic seizures
- Dysmorphic features– facial bone abnormalities, underdeveloped nails (nail hypoplasia), finger and toe abnormalities (brachytelephalangy) and abnormal genital-urinary tract features.
- Gastrointestinal– anal deformities, and intestinal abnormalities (Hirschsprung disease)
Other reported symptoms of PIGO-CDG include hearing loss and stunted growth. Less common symptoms include dry, thickened skin (keratoderma), renal abnormalities and heart defects1,5.
Biochemical Abnormalities
Biochemical abnormalities observed in individuals with PIGO-CDG include elevated alkaline phosphatase levels. Alkaline phosphatase is a GPI-anchor protein that is normally found on the cell surface but may be secreted as a soluble protein in PIG deficiencies.
Classification
PIGO-CDG is classified as a disorder of GPI-anchor biosynthesis.
Diagnosis
GPI-related CDG should be considered in individuals presenting with early onset severe seizure disorders and dysmorphic facial features, even if transferrin and total N-glycan analysis are normal. As currently available screening tests for CDG will not reliably detect PIGO-CDG, diagnosis is typically achieved through genetic testing, either as part of an epilepsy panel or whole exome sequencing. PIGO-CDG may also be primarily screened for by analyzing surface GPI-anchored proteins on blood cells by flow cytometry.
GPI-Anchored Protein Flow Cytometry
Individuals with PIGO-CDG lack GPI-anchored proteins on the surface of their granulocytes.
Prognosis
The prognosis of PIGO-CDG may vary depending on the severity of an individual’s symptoms. The broad clinical spectrum may result in premature death in some patients. In the 18 reported cases, 6 deaths have been reported in patients ranging from 9 months old to 2.7 years of age5,6 6,7. As of 2017, the oldest reported patient was 22 years old1.
Management
Management of symptoms in individuals with PIGO-CDG may include combinations of physical therapy, occupational therapy, and palliative measures. In some patients, seizures are treatable with anti-epileptic medication5.
Therapies
There are currently no treatment options available for PIGO-CDG, however, a homology-independent strategy for targeted transgene integration (HITI) gene therapy was investigated in mouse models of PIGO deficiency study9.
Pyridoxine, a form of vitamin B6, has been investigated as a treatment for seizures in PIGO-CDG, but its efficacy is unclear 4,15. As pyridoxine has only been evaluated in three patients with PIGO-CDG, a further clinical investigation is needed to determine its efficacy in this CDG type. Pyridoxine has been investigated as a treatment for GPI disorders because it is thought that there may be a reduced level of intracellular pyridoxine, specifically in the brain, due to disrupted GPI-anchored protein function15.
Research Models
Several PIGO mouse models have been generated to study PIGO-CDG. Research models are available to study PIGO-CDG in mice.
Mouse (M. Musculus)
Pigo-/- constitutive knockout
Homozygous Pigo constitutive knockout mice are embryonic lethal prior to organogenesis (E9.5) (IMPC).
Pigo+/- heterozygous knockout
Heterozygous Pigo knockout mice display an increased heart rate and a cardiovascular phenotype by ECG (IMPC).
Homozygous Pigo Arg119Trp/Arg119Trp knock-in
In PIGO-CDG patients, the p.Arg119Trp variant results in a significant decrease in enzyme activity and severe clinical symptoms7, however, in mice the variant is mild 9. Homozygous Pigo Arg119Trp knock-in mice displayed a susceptibility to seizures and poor cognitive function9.
PigoArg119Trp/- knock-in/knockout
Pigo knock-in/knockout mouse (KIKO) models in which one allele is a knock-in Pigo missense variant and the other allele is a Pigo knockout, mimics the genotype of PIGO-CDG patients with compound heterozygous bi-allelic PIGO mutations. Pigo Arg119Trp/- KIKO mice displayed a normal lifespan, an onset of tremors at 7-16 weeks, severe defects in muscle coordination and considerable susceptibility to seizures with poor cognitive function9.
Homozygous Pigo Thr130Asn/Thr130Asn knock-in
The p.Thr130Asn variant results in a severe phenotype in mice. Pigo Thr130Asn/Thr130Asn knock-in mice displayed a decreased expression of GPI-APs on blood granulocytes, an increased susceptibility to seizures, early onset (6 weeks) and increased frequency of tremors, decreased growth and survival (median survival ~ 40 weeks), hyperphosphatasia, and ataxia. An AAV-based gene therapy was evaluated in this mouse model which resulted in improvements in growth, susceptibility to seizures, muscle strength and coordination, and expression of surface GPI-APs9.
PigoThr130Asn/- knock-in/knockout
Pigo Thr130Asn/Thr130Asn KIKO mice display a decreased expression of GPI-APs on blood granulocytes, increased susceptibility to seizures, early onset (5 weeks) and increased frequency of tremors, decreased growth and survival (median survival ~15 weeks), hyperphosphatasia, ataxia and reduced muscle volume. Similar to the clinical presentation in PIGO deficiency patients, mice also have a thin corpus callosum and anterior commissure by brain MRI. An AAV-based gene therapy was evaluated in this mouse model which resulted in significant improvements in growth, susceptibility to seizures, muscle strength and coordination, and expression of surface GPI-Aps9.
Homozygous Pigo Lys1051Glu/Lys1051Glu knock-in
The Pigo missense variant p.Lys1051Glu in mice is analogous to the p. Lys1047Glu variant in PIGO-CDG patients. Pigo Lys1051Glu/Lys1051Glu knock-in mice displayed normal survival times, no tremors, and no susceptibility to spontaneous seizures, but presented with epileptic discharges via EEG measurement9.
Pigo Lys1051Glu /- knock-in/knockout
PigoLys1051Glu /- KIKO mice display normal survival times, no tremors, and no susceptibility to spontaneous seizures9.
Clinical Studies
Active
Clinical and Basic Investigations into Congenital Disorders of Glycosylation (NCT04199000)
The Frontiers in Congenital Disorder of Glycosylation Disorders Consortium (FCDGC) is conducting a 5-year natural history study on all CDG types, including PIGO-CDG. The purpose of this study is to define the natural history and clinical symptoms of CDG, develop new diagnostic techniques, identify clinical biomarkers that can be used in future clinical trials and evaluate whether dietary treatments improve clinical symptoms and quality of life.
Organizations
GPI-anchor CDG Community Facebook Group
Publications
PIGO-CDG Scientific Articles on Pubmed
Additional Resources
Kinoshita Lab (GPI anchor pathway researchers)
IEMbase
OMIM
Orphanet
MedlinePlus
Genetic Testing Registry
ClinVar
NIHGeneCards
UniProt
Marrvel
References
- Morren, M.A. et al. PIGO deficiency: palmoplantar keratoderma and novel mutations. Orphanet J. Rare Dis. 12, 101 (2017).
- Xue, J., Li, H., Zhang, Y. & Yang, Z. Clinical and genetic analysis of two Chinese infants with Mabry syndrome. Brain Dev. 38, 807–818 (2016).
- Nakamura, K. et al. PIGO mutations in intractable epilepsy and severe developmental delay with mild elevation of alkaline phosphatase levels. Epilepsia 55, (2014).
- Kuki, I. et al. Vitamin B6-responsive epilepsy due to inherited GPI deficiencyichiro. Neurology vol. 81 1467–1469 (2013).
- Holtz, A. M. et al. Expanding the phenotypic spectrum of Mabry Syndrome with novel PIGO gene variants associated with hyperphosphatasia, intractable epilepsy, and complex gastrointestinal and urogenital malformations. Eur. J. Med. Genet. 63, 103802 (2020).
- Zehavi, Y. et al. A homozygous PIGO mutation associated with severe infantile epileptic encephalopathy and corpus callosum hypoplasia, but normal alkaline phosphatase levels. Metab. Brain Dis. 32, 2131–2137 (2017).
- Tanigawa, J. et al. Phenotype-genotype correlations of PIGO deficiency with variable phenotypes from infantile lethality to mild learning difficulties. Hum. Mutat. 38, 805–815 (2017).
- Krawitz, P. M. et al. Mutations in PIGO, a member of the GPI-anchor-synthesis pathway, cause hyperphosphatasia with mental retardation. Am. J. Hum. Genet. 91, 146–151 (2012).
- Kuwayama, R. et al. Establishment of mouse model of inherited PIGO deficiency and therapeutic potential of AAV-based gene therapy. Nat. Commun. 13, 3107 (2022).
- Kinoshita, T. Biosynthesis and biology of mammalian GPI-anchored proteins. Open Biol. 10, 190290 (2020).
- Kinoshita, T. & Fujita, M. Biosynthesis of GPI-anchored proteins: Special emphasis on GPI lipid remodeling. Journal of Lipid Research vol. 57 6–24 (2016).
- Liu, Y. S. & Fujita, M. Mammalian GPI-anchor modifications and the enzymes involved. Biochemical Society Transactions vol. 48 1129–1138 (2020).
- Hong, Y. et al. Requirement of PIG-F and PIG-O for transferring phosphoethanolamine to the third mannose in glycosylphosphatidylinositol. J. Biol. Chem. 275, 20911–20919 (2000).
- Fujita, M. & Kinoshita, T. Structural remodeling of GPI anchors during biosynthesis and after attachment to proteins. FEBS Lett. 584, 1670–1677 (2010).
- Tanigawa, J. et al. High-dose pyridoxine treatment for inherited glycosylphosphatidylinositol deficiency. Brain Dev. 43, 680–687 (2021).
Show More
